
|
Lazy eyes listen
|
- Over 1200 deaths were reported in the first 90 days
- The report was wrestled out by FOIA and has been avoided by mainstream
- Pfizer labeled the data “proprietary and confidential” which is “weird” for safety data on EUA product which should be readily public
- “There is an overwhelming tilt towards women reporting Adverse Events often on the order of 4x or more”
by Chris Martenson, PeakProsperity
This episode reviews a previously hidden report by Pfizer to the FDA covering the first 90 days of ‘post authorization’ vaccine safety data. A judge ordered its release and, perhaps not surprisingly, no major news outlet has dared to cover the story.
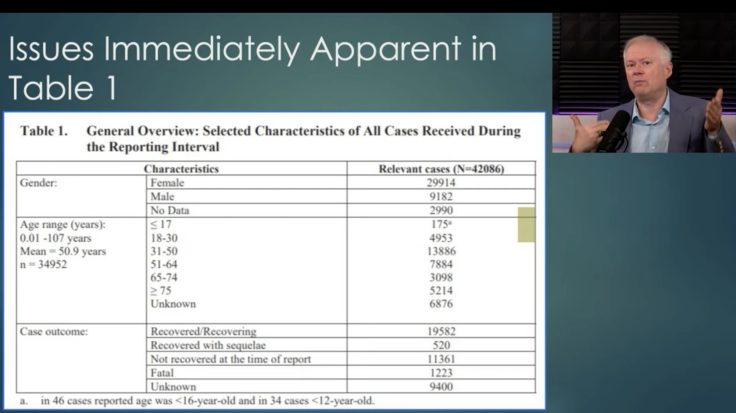
Within that first 90- day window, over 1,200 deaths were reported, with a significant number appearing to happen within the first 24 hours after vaccination. We can’t say for sure because the report lacks critical details that would allow us to align the specific deaths with the reported observation that the median elapsed time between vaccination and the adverse event was “<24 hours” for many types of AEs.
This report is a ‘second go’ by Pfizer after the first report was deemed wholly insufficient and lacking detail. This one isn’t actually all that much better, as it is entirely passive (no active data collection undertaken – it relies entirely on “spontaneously” reported events, and no inquiry into the adverse events is part of this version of the report), there’s no attempt made to define the incidence of events, and there is no visible effort made to compare the levels of events to an expected baseline of such events.
It is also impossible to determine first vs second vaccination injections (not reported) and the age brackets are not even age brackets (“child, adult and elderly”).
Besides the 1,223 spontaneously reported deaths, there is an overwhelming tilt towards women reporting AE’s often on the order of 4x or more. None of these were deemed to be worthy of modifying either the administration of the vaccines or the collection of new data in more useful detail.
Further, I track down the actual state of knowledge of what is and is not known about pregnancy and the vaccines to uncover the fact that no providers can say, one way or the other, if the vaccines are safe.
In the most current language available to vaccinators and health care providers, Pfizer says “Available data on COMIRNATY administered to pregnant women are insufficient to inform vaccine-associated risks in pregnancy.”
Naturally, without being able to articulate the actual risk, true informed consent is not possible for pregnant women seeking to understand what the risks might be to their unborn child. All that can truthfully be said is “we don’t know.”
If more robust data does exist, it is not yet been made public. We may have to wait up to 55 years for that.
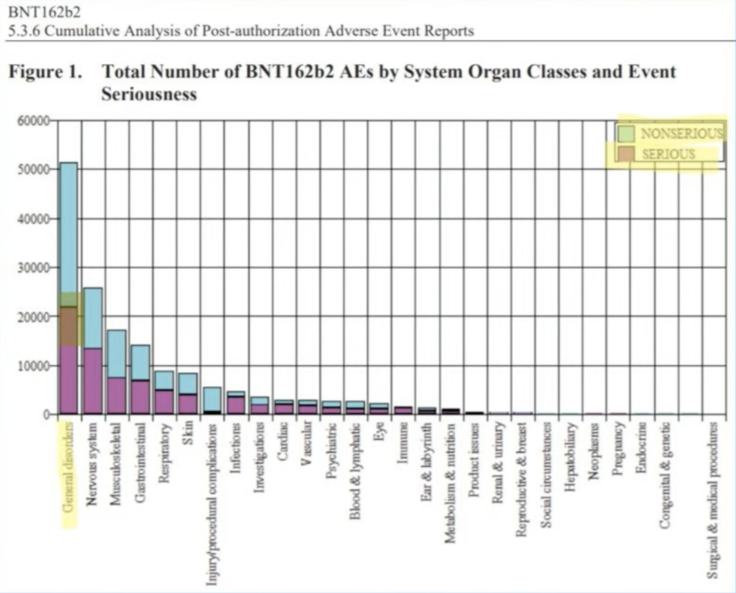
Report title: 5.3.6 CUMULATIVE ANALYSIS OF POST-AUTHORIZATION ADVERSE EVENT REPORTS OF PF-07302048 (BNT162B2) RECEIVED THROUGH 28-FEB-2021
Links to Referenced Material
- Mattias Desmet interview
- AMA – Informed Consent
- Informed Consent – Pfizer Proivder Labelling (Pregnancy)
FAIR USE NOTICE This video may contain copyrighted material; the use of which has not been specifically authorized by the copyright owner. We are making such material available for the purposes of criticism, comment, review and news reporting which constitute the fair use of any such copyrighted material as provided for in section 107 of the US Copyright Law. Not withstanding the provisions of sections 106 and 106A, the fair use of a copyrighted work for purposes such as criticism, comment, review and news reporting is not an infringement of copyright.
This video is intended for EDUCATIONAL and ENTERTAINMENT purposes ONLY and is NOT to be construed as LEGAL, FINANCIAL or MEDICAL ADVICE. Repeat: THIS IS NOT LEGAL, FINANCIAL or MEDICAL ADVICE. We are not legal, financial or medical experts. In case we lose our YouTube channel, be prepared to subscribe to us in other ways.
Episode 038
00:00 – Intro
02:17 – Pfizer Safety Data “Leaked”
09:38 – Known Limitations of SAEs
12:59 – Issues Immediately Apparent in Table 1
21:32 – Safety: Preganancy
24:08 – Children – Off Label Use
27:09 – Heart Issues (Known by Feb 28, 2021)
31:15 – Immune-Mediated
33:34 – Informed Consent
40:44 – What is “informed Consent” for Pregnancy?
43:00 – Conclusions




